31 0.1 ml 0.001 M hcl solution is diluted with water to make 10 litres .calculate PH of the dilute solution.

Reagents and conditions: (a) 10% NaOH, MeOH, reflux or 0.1 M HCl-EtOH,... | Download Scientific Diagram
What will be the concentration of CH3COO– in 0.1 M HCl solution, if dissociation cons†an t of 0.01 M CH3COOH is 1.8 × 10–5?

CANLI KİMYASI LABORATUVARI NO: 3 ÇÖZELTİ HAZIRLAMA VE KONSANTRASYON KAVRAMLARI Araş. Gör. Gökçe TANER. - ppt indir

The volume of 0.1M HCl required to neutralize completely 1g of an equimolar mixture of {Na}_{2}{CO}_{3} is:318.76mL215mL325mL157mL
nt20 ml of 0.1 M solution of compound Na2CO3.NaHCO3.2H2O is titrated against 0.05M HCl, x mL of HCl is used when phenolphtalein is used as an indicator and y mL of HCl
To a 50 ml of 0.1 M HCl solution, 10 ml of 0.1 M NaOH is added and the resulting solution is diluted to 100 ml. - Sarthaks eConnect | Largest Online Education Community

227 50 ml of 0.1 M HCl is mixed with 50 ml of 0.05 M H2SO4, then the resultant (CH) in the solution 6ook ve 300m 0.05 M (3) 0.1 M (2) 0.025 M (4) 0.5 M ocem
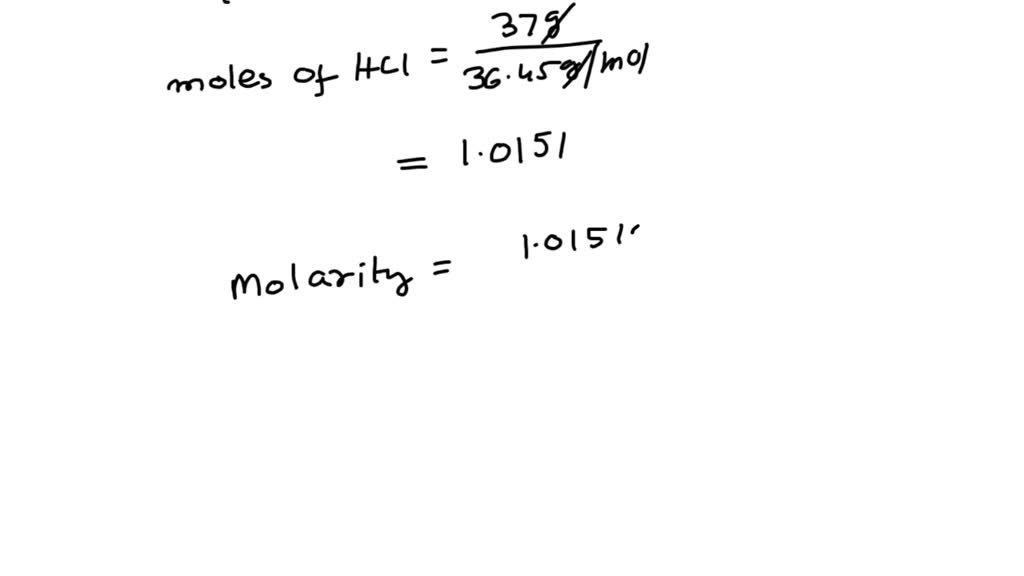
SOLVED: How can you prepare 500 mL 0.1 M HCl solution from a HCl that is 37% (w/v). Indicate how many mL of HCl is required. Show your calculations

L-Lysine Hydrochloride Amino Acid 100mM 0.1 M HCl - Laboratory Research Solution | Procurenet Limited

SOLVED: Prepare 30 mL of 0.1 M HCl from HCl stock solution: V (0.2 mL) (ia) V2 M1V1 = M2V2 30 mL = V2 Prepare 50 mL of 0.25 mg/mL DTT from















