These highlights do not include all the information needed to use FERRIPROX safely and effectively. See full prescribing information for FERRIPROX. FERRIPROX® (deferiprone) tablets, for oral useInitial U.S. Approval: 2011
KISA ÜRÜN BİLGİSİ 1. BEŞERİ TIBBİ ÜRÜNÜN ADI FERRİPROX 500 mg film kaplı tablet 2. KALİTATİF VE KANTİTATİF BİL

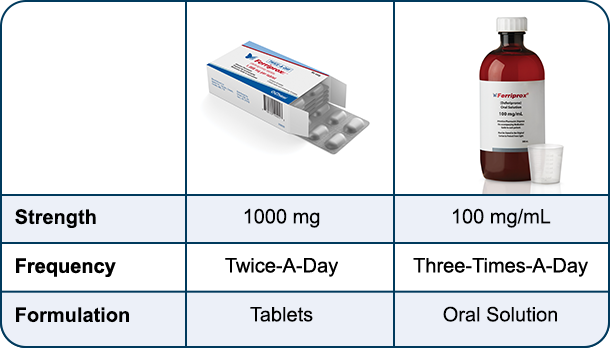
Thalassaemia International Federation - TIF - U.S. Food and Drug Administration (FDA) approved a new formulation for Ferriprox (Deferiprone) twice-a-day tablets, suitable for the treatment of #patients with transfusional iron overload due