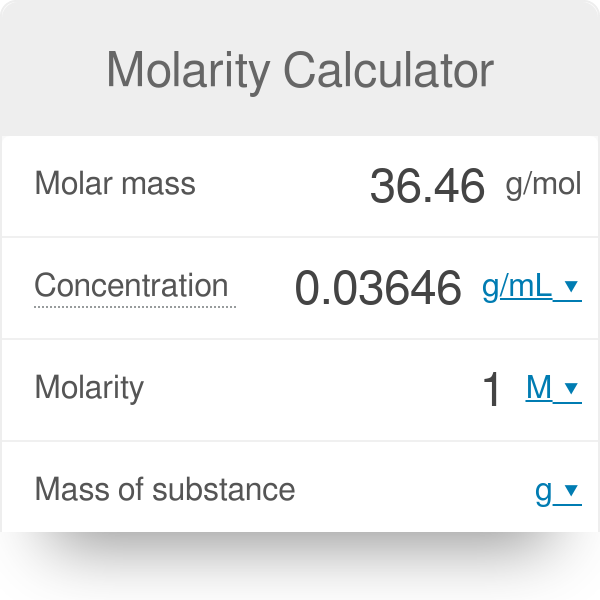
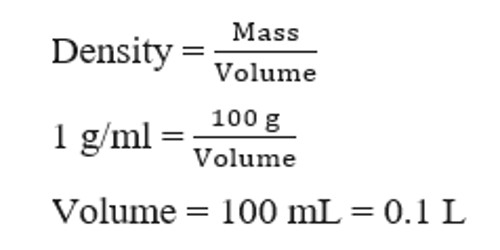Molarity 2. Molarity (M) this is the most common expression of concentration M = molarity = moles of solute = mol liters of solution L Units are. - ppt download

Calculating molarity units molar concentration of solutions practice questions on molarity how to make up a standard solution how to determine solubility of a salt gcse chemistry igcse KS4 science A level

Molarity . . Molar concentration (also called molarity, amount concentration, or substance concentration) is a measure of the concentrati... | Instagram

Molar concentration of M +/2+ ions left free by InsP 6 at different pH... | Download Scientific Diagram

Calculating molarity units molar concentration of solutions practice questions on molarity how to make up a standard solution how to determine solubility of a salt gcse chemistry igcse KS4 science A level

SOLVED: Determine the molar concentration of the dye in the stock dye solution that you used, as follows: (show work for credit) Read the absorbance value (AO) at the peak of the